Conjugated compounds
Conjugated compounds
Aromatic compounds
•Benzene causes a depressed white blood cell count on prolonged exposure and should not be used as a laboratory solvent
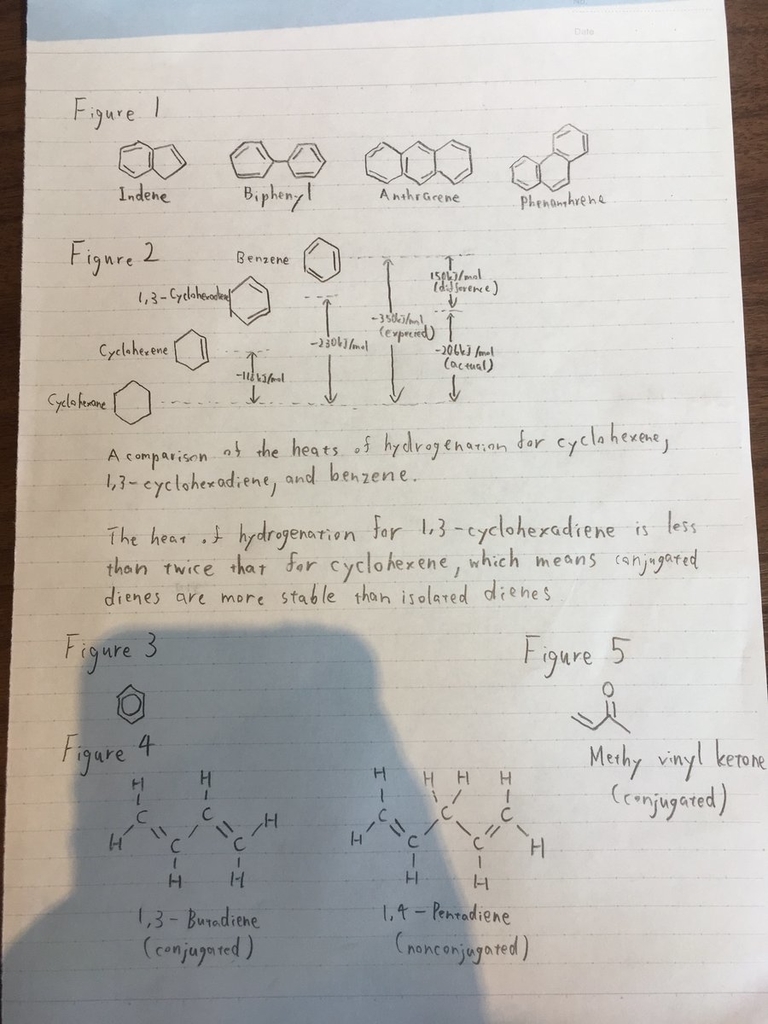
•Fractional distillation of coal tar yields benzene, toluene, xylene, naphthalene, and a host of other aromatic compounds such as Figure 1
•Unlike coal, petroleum contains few aromatic compounds and consists largely of alkanes. During petroleum refining, however, aromatic molecules are formed when alkanes are passed over a catalyst at about 500℃ under high pressure.
•benzene’s stability can be explained by measuring heats of hydrogenation(see Figure 2 for more information)
•benzene is sometimes represented like figure 3. however, it doesn’t indicate the number of π electrons in the ring.
1,3-Butadiene(see Figure 4)
•this is the example of compounds that have different unsaturations close together which interact with one another.
•1,4-Pentadiene is nonconjugated(see Figure 4)
enones
•an enone is a type of organic compound consisting of an alkene conjugated to a ketone.
•progesterone, the hormone that prepares the uterus for implantation of a fertilized ovum is also a conjugated enone
•the simplest enone is methyl vinyl ketone(see figure 5)
conjugated polyene
•lycopene, the red pigment found in tomatoes and thought to protect against prostate cancer, is a conjugated polyene